About Us
Founded in 2009, Genesys Spine® is a rapidly growing spinal fusion device company based in Austin, Texas. As a privately held company, we pride ourselves in being able to adapt to the needs of our surgeons and make decisions quickly.
As a result, we have successfully released over twenty FDA-cleared product lines with several other products in various stages of the design and development cycle.
We are proud of our company, the success we have achieved, and the reputation we have earned with our customers. Our goal of creating market leading technology is driven through engineering excellence, unparalleled support, and surgeon efficiency. These three elements have become the principles of the company as we strive to create exceptional products and trusting, responsible relationships.
Engineering Excellence
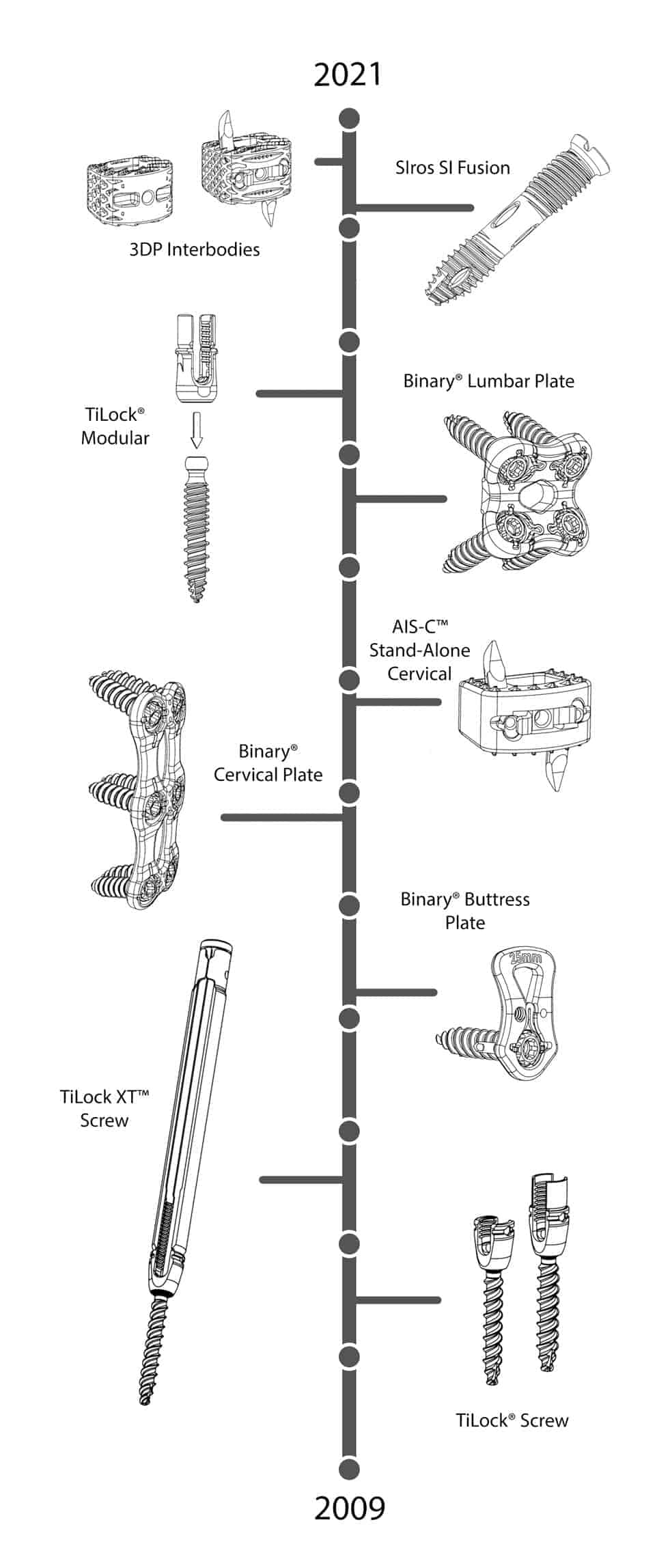
 Engineering excellence takes years of dedication, industry specialization and innovative thinking. Some of our most noteworthy engineering innovations include:
Engineering excellence takes years of dedication, industry specialization and innovative thinking. Some of our most noteworthy engineering innovations include:
- First to market, 3D Printed non-screw based, zero-profile, direct anterior standalone interbody system for the cervical spine
- First of its kind, Peek non-screw based, zero-profile, direct anterior standalone interbody system for the cervical spine
- First of its kind, 3D printed threaded implants intended to treat degenerative sacroiliitis and sacroiliac joint disruptions.
- Patented zero-step locking mechanisms on our Pedicle Screw, Buttress, Lumbar, and Anterior Cervical Plates
- Proprietary Opposing Angle Interface (OAI) Set Cap

Unparalleled Support
As a team, our highest priority is to provide outstanding service and unparalleled support to our customers. We are dedicated to providing surgeons a product that excels in the competitive landscape, reduces surgical time and effort, and ultimately minimizes hospital stays and recovery times.
Surgeon Efficiency
 Our goal is to ensure surgeon efficiency in the operating room. Our products and instruments are designed to ensure consistent and reproducible results every time. The results are streamlined products that improve surgeon’s speed, efficiency, and effectiveness.
Our goal is to ensure surgeon efficiency in the operating room. Our products and instruments are designed to ensure consistent and reproducible results every time. The results are streamlined products that improve surgeon’s speed, efficiency, and effectiveness.
Our products, which include proprietary features and system specific instruments, have earned multiple awards, garnered positive feedback from the marketplace, and have remained within existing parameters for today’s current reimbursement codes.
Our entire team is focused on the quality and usability of our products. We are honored to provide our customers with solutions that meet their needs and most importantly, improve the welfare of their patients.
The Genesys Spine flagship product, the TiLock® Pedicle Screw System, was introduced in August of 2010 and has continued to receive excellent reviews from the surgeon community for its ease of use and intuitive characteristics.
The AIS-C® Standalone System, a first of its kind, non-screw based, zero-profile, direct anterior standalone interbody system for the cervical spine launched in 2018 and has received multiple design awards including a Gold Medical Design Excellence Award. Recently, we announced the launch of the 3D Printed AISC-C Standalone System. This is the first 3D printed non-screw based cervical standalone cage in the United States.
The SIros™ Sacroiliac Joint Fusion System is the very first 3D Printed system to launch in the US. With a goal of reducing operative times, lowering surgical complications, and improving clinical outcomes, the system was developed with a novel feature for cannulated bone screws that enables bone fragments to move through the screw’s cannula without binding. This was accomplished by incorporating a rifled inner cannula into the screw. Specifically, the rifling is comprised of raised helical ridges (flutes) that are 3D printed into the internal (bore) surface of the screws.
Our TiLock® Modular Spinal System uniquely addresses the challenges of posterior fixation by providing surgeons with the flexibility to secure difficult operative levels. The system received an Iron A Bronze Award for its innovative design.
Our Promise
Genesys continues to work diligently toward the development and release of new product lines and to provide the market with quality products, service, and support.
To learn how we can satisfy your surgical needs, please contact us.